Have you ever wondered what makes up everything around you, from the air you breathe to the food you eat?
What if I told you that every single thing in the universe is made of just a handful of building blocks, and they all fit together on a special chart?

Key Takeaways
Welcome to the fascinating world of the periodic table of elements! This incredible chart isn’t just a collection of symbols and numbers; its your gateway to understanding the elements that make up our universe.
It’s like a secret code that unlocks the mysteries of chemistry, helping you discover how elements interact and why they behave the way they do.
How they react with other elements combine and form the substances that make up everything in our lives.
Whether you’re just starting your chemistry journey studying GCSE combined-science or looking to uncover more about the elements, this blog post will guide you through the basics of the periodic table
At first, it might seem complex, but with this simple guide, you will start to learn the basics and get a true understanding of the periodic table.
What Is the Periodic Table?
The periodic table is like a map of all the known elements in the universe.
It organizes elements based on their atomic number (the number of protons in an atom’s nucleus, which is in the centre of the atom) and shows us how different elements interact and behave.
It’s not just a list; it’s a clever chart. It helps predict what elements will be like based on where they are.
What is an element?
Chemical elements are pure substances made of one type of atom.
The atomic structure includes protons, neutrons, and electrons.
Protons have a positive charge, neutrons are neutral, and electrons are negative.
The number of protons in the nucleus sets each element apart. This is why each element on the Periodic Table is different.
The History of the Periodic Table?
The story of the periodic table is both fascinating and complex. It began centuries ago with early attempts to sort elements. Dmitri Mendeleev’s work in the 19th century was a major breakthrough. This section explores the key moments in the history of the periodic table.

Early Attempts to Organise Elements
In the early 19th century, people started trying to group elements. Johann Wolfgang Döbereiner found ‘triads’ in 1829. He noticed that elements like chlorine, bromine, and iodine had similar properties.
Later, John Newlands came up with the Law of Octaves in 1865. He said that every eighth element had similar traits when listed by atomic mass.
Chemists like Antoine Lavoisier and Stanislao Cannizzaro also played a big role. By 1860, the first chemistry conference in Karlsruhe, Germany, set hydrogen atom’s atomic weight to 1.
This helped lay the groundwork for the periodic table.
Mendeleev’s Contribution and Later Developments
In 1869, Dmitri Mendeleev published his periodic table.
He organized the 63 known elements according to their atomic weight and expected the discovery of new elements.
He left gaps in his table for elements like eka-aluminum and eka-silicon.
‘eka’ means ‘one’ (one beyond) – ‘eka-aluminium’ meaning ‘the one beyond aluminium,’ which at the time was unknown.
These predictions were later confirmed with the discovery of gallium in 1875 and germanium in 1886. Mendeleev’s work was a huge leap forward in chemistry.
He and Lothar Meyer were awarded the Davy Medal in 1882. This recognition highlighted their groundbreaking contributions to chemistry.
| Year | Event |
|---|---|
| 1860 | The first international conference of chemistry in Karlsruhe established hydrogen’s atomic weight as 1. |
| 1865 | John Newlands proposed the “Law of Octaves.” |
| 1869 | Dmitri Mendeleev published his periodic table, predicting properties of undiscovered elements. |
| 1875 | Discovery of gallium, validating Mendeleev’s predictions. |
| 1882 | Mendeleev and Lothar Meyer awarded the Davy Medal. |
| 1886 | Discovery of germanium, further validating Mendeleev’s predictions. |
| 1913 | Henry Moseley rearranged the periodic table by atomic number using X-ray wavelengths. |
| 2019 | UNESCO celebrated the 150th anniversary of the periodic table with the International Year of the Periodic Table. |
In 1913, Henry Moseley created a new version of the periodic table to use atomic number instead of weight.
He used X-ray wavelengths for this. This update made the table more accurate and aligned with modern atomic theory.
The Basic Structure of the Periodic Table
The periodic table is a key tool that shows trends and properties of elements. It does this based on their atomic mass and structure.
To understand these trends, we need to look at how elements are arranged in the table.
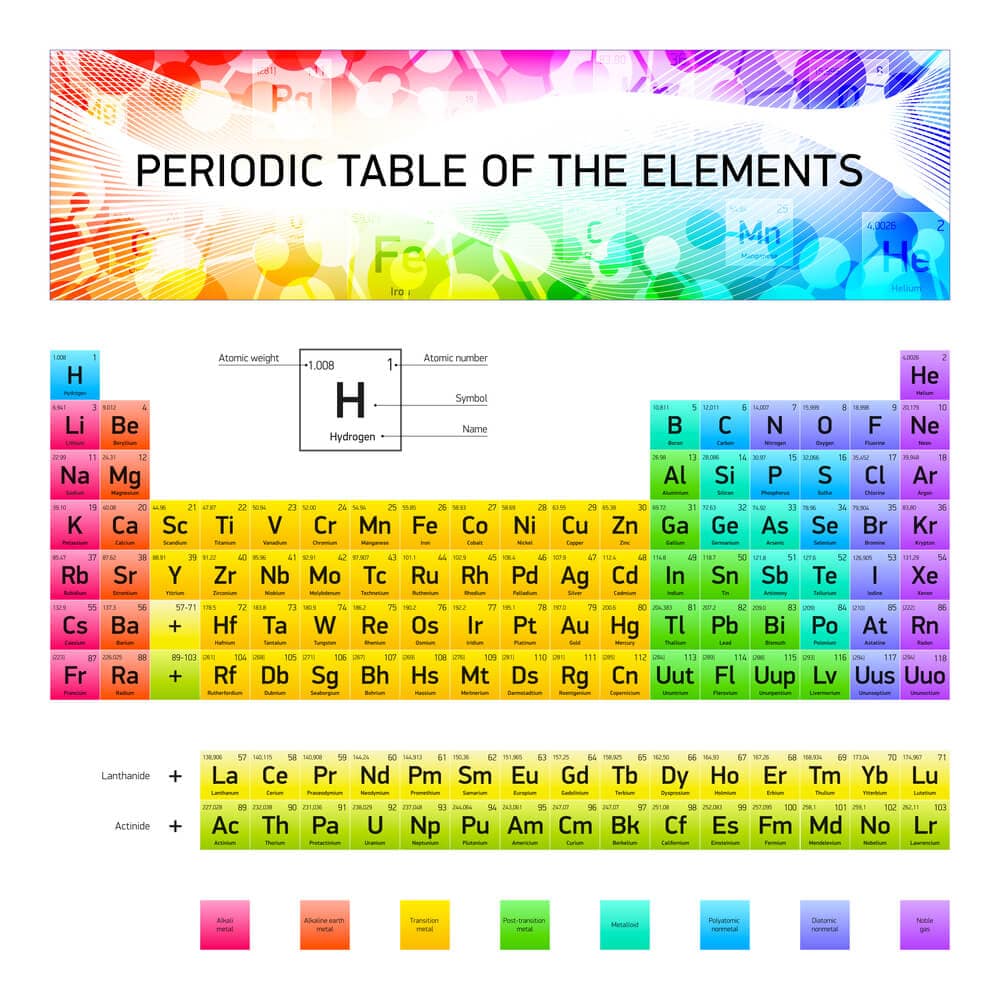
Element symbol
Each element has a unique symbol made up of one or two letters, with the first letter always capitalized.
For example, H stands for hydrogen and Ca stands for calcium.
This method helps identify elements and shows how they are connected in the field of chemistry
Atomic Number and Atomic Mass
The heart of the periodic table is atomic number and atomic mass.
Each element’s spot is based on its unique atomic number; the atomic number is the number of protons in the nucleus of the element’s atoms.
The atomic number is important because it tells us exactly what the element is.
For example, oxygen has an atomic number of 8, telling you it has 8 protons; no other elements will have 8 protons in their nucleus; they will either have more or fewer protons.
The elements are arranged in order of increasing atomic number from left to right.
This arrangement forms the basis of the periodic law, leading to trends in properties like reactivity and ionisation energy. For instance, as the atomic number increases, the atomic mass generally increases as well
The atomic mass number is the total number of protons plus the number of neutrons in an atom’s nucleus. It is measured in atomic mass units (amu), giving a fuller picture of an element’s weight.
By looking at these numbers, we can see trends that help predict element properties.
Groups and Periods
The periodic table is organised into vertical columns called groups and horizontal rows known as periods.
There are 18 groups numbered from left to right. Elements in the same group share similar chemical properties due to the number of electrons in their outer shell (also called valence shells).
For instance, Group 1 includes alkali metals like lithium and sodium, which are highly reactive.
There are seven periods in the table, numbered from top to bottom. reflect the number of electron shells that an element possesses.
For example, lithium, located in period 2, has two electron shells, while sodium, located in period 3, has three.
| Period | Electron Shells | Number of Elements |
|---|---|---|
| Period 1 | 1 | 2 (H,He) |
| Period 2 | 2 | 8 |
| Period 3 | 3 | 8 |
| Period 4 | 4 | 18 |
| Period 5 | 5 | 18 |
| Period 6 | 6 | 32 |
| Period 7 | 7 | 32 |
As you move from left to right across a period, properties of elements change.
For example, the first period contains only hydrogen and helium, while the second period includes elements like lithium and neon.
Electron Configuration and Periodic Trends
Electron configuration is key to understanding an element’s chemical behaviour. These electrons are arranged in shells and tell us how elements react.
Trends like atomic radius and electronegativity change in a pattern, giving us clues about how elements bond and react.
Categories of Elements: Metals, Non-Metals, and Metalloids
The periodic table is divided into three main categories: metals, nonmetals, and metalloids.
These categories help you predict the behaviour and reactions of different elements.
Importance of the Periodic Table in Chemistry Education
The periodic table serves as a foundational tool for understanding the chemical properties and behaviours of elements, making it essential for students studying chemistry.
Familiarity with its layout is particularly critical for GCSE examinations, where a fresh copy of the periodic table is provided to students.
Different examination boards, such as AQA, Edexcel, and OCR, may present slight variations in the periodic table, including group numbering and placement of elements, which can affect how students interpret and use the table during exams
Key Features for GCSE Students:
– Electronic Configurations: Understanding the electronic configurations of the first 20 elements is crucial for predicting chemical behavior.
– Melting and Boiling Points: These properties help us understand the states of matter and physical changes.
– Metals vs. Non-Metals: The table clearly distinguishes between metals, non-metals, and metalloids, each with unique properties.
Practical Applications:
– Predicting Properties and Reactions: The periodic table is a powerful tool for predicting how elements will interact and react with each other.
– Real-World Uses: From the helium in balloons to the silicon in computer chips, the elements have countless applications in our daily lives.
Explore Online: Dive into interactive periodic tables online to discover more about each element.
Here is a great Interactive Periodic Table to use; just click this link RSC Interactive Periodic Table
Fun Facts About Elements:
Conclusion
The periodic table is more than just a chart; it’s a gateway to understanding the universe’s building blocks, playing a critical role in chemistry and other sciences.
We’ve explored the periodic table’s structure, history, and trends. It’s a vital tool in education and science. The table has 7 periods and 18 groups, showing how elements are arranged by atomic number.
This arrangement helps us understand chemical behaviour. It lets us predict an element’s properties and how it reacts.
Elements like calcium and sodium show the table’s real-world use. They are found in everyday things and living things. The table also helps us learn about rare elements.
For students and teachers, the periodic table is essential. It helps build a strong science base. By learning about it, we can explore chemistry and material science more.
If you’re eager to learn more about the periodic table and chemistry, check out our YouTube channel for engaging chemistry content
Join our community on Facebook to ask questions as well as share your own discoveries.
FAQ
The periodic table is a way to list chemical elements. It groups them by their properties and atomic structure. This makes it easy for scientists and students to find information.
Elements are listed by their atomic numbers, which tell us how many protons they have. The table has rows and columns. Elements in the same column have similar properties because of their electron arrangements.
Periods are the rows in the table that show how properties change. Groups are the columns where elements have similar traits. This is because of their electron setups.
A chemical element is defined by its atomic number, which is the number of protons in its nucleus. Elements are pure substances made of only one type of atom. This sets them apart from other substances.
Protons and neutrons are in the nucleus and have positive and neutral charges. Electrons, with a negative charge, orbit around the nucleus. The number of protons tells us what element it is. The way electrons are arranged affects its chemical behaviour.
The periodic table started with early attempts to classify elements. Dmitri Mendeleev’s 1869 table organised them by atomic weight. Later, it was updated to use atomic numbers for better accuracy.
Dmitri Mendeleev was a Russian chemist who created the first widely accepted periodic table in 1869. He sorted elements by atomic weight and predicted the existence and properties of new elements.
Atomic mass is the total number of protons and neutrons in an atom. The atomic number is just the number of protons. These values tell us about an element’s properties and where it goes in the periodic table.
Electron configuration shows how electrons are arranged around the nucleus. It affects an element’s chemical behaviour and trends like electronegativity and atomic radius.
lements are divided into metals, non-metals, and metalloids. Metals are conductive and can be shaped easily. Non-metals have different forms and are not conductive. Metalloids have properties of both metals and non-metals.
Element symbols are short names for each element. The atomic number is above the symbol, and the atomic mass number is below. This helps identify the element quickly.
Colour codes and legends in the periodic table help identify element types like alkali metals and noble gases. These visual aids make it easier to understand and use the table.



2 Comments
ARIKIRIZA ASTON · 03/09/2026 at 4:52 AM
Good work 👏
Rodney · 03/09/2026 at 7:28 PM
Thank you 😊