Introduction
Every time you step into the kitchen, you’re not just preparing a meal—you’re delving into the captivating chemistry of cooking!
The chemistry of cooking blends art and science seamlessly, transforming raw ingredients into delectable dishes.
By understanding the chemical reactions happening in your kitchen, you can unlock the secrets to elevating your culinary skills and creating truly flavourful meals. Let’s delve into the world of cooking chemistry and discover how it can transform your kitchen creations!
The Kitchen: Your Personal Chemistry Lab
Think of your kitchen as a laboratory where you mix ingredients, apply heat, and observe changes. Cooking is all about chemistry, the science of how substances interact and transform.
Essential Chemical Components in Everyday Cooking
Ingredients like proteins, carbohydrates, fats, and vitamins are key. They affect the taste, texture, and result of your dishes. Learning how these essential chemical components work is crucial for great cooking.
Basic Kitchen Equipment for Chemical Reactions

From simple pots and pans to special utensils, your kitchen tools help with chemical reactions. Knowing how each piece works can make you better at kitchen chemistry.
| Kitchen Equipment | Chemical Reaction Role |
|---|---|
| Pots and Pans | Facilitate heat transfer and temperature control for chemical reactions |
| Whisks and Mixing Bowls | Promote even distribution and integration of ingredients for chemical interactions |
| Baking Sheets and Trays | Enable controlled heating and controlled evaporation for browning and caramelisation |
By getting into kitchen chemistry and culinary science, you can improve your cooking. This way, you can make dishes that truly stand out.
Chemistry of Cooking: From Raw to Transformed
Cooking is a magical journey where raw ingredients turn into delicious dishes. This happens through complex chemical processes. Heat, acids, and enzymes work together in the kitchen to create tasty meals.
For example, marinating chicken in lemon juice makes it tender. This is because the acidity breaks down the proteins. Chefs use this knowledge to make food taste and feel just right.
The Maillard Reaction and Caramelisation: Creating Flavour and Colour
Two important chemical reactions in cooking are the Maillard reaction and caramelisation:

Caramelisation: Caramelisation, on the other hand, happens when sugar is heated to a high temperature, breaking down into a range of flavourful compounds. This process gives caramel its rich, sweet flavour and is responsible for the golden colour of roasted vegetables and baked goods.
| Chemical Process | Culinary Application | Transformation |
|---|---|---|
| Maillard Reaction | Baking Bread Crusts | Flavour Compounds, Golden-brown Hues |
| Protein Denaturation | Marinating Chicken | Tenderised Meat |
| Caramelisation | Crème Brûlée Topping | Altered Flavour and Colour |
The Role of Heat in Cooking
Heat is a powerful tool in the kitchen, changing the structure and taste of food.
Here are a few ways heat transforms our meals:
Starch Gelatinisation: When you cook pasta or rice, the heat causes starch granules to absorb water and swell, making them soft and palatable. This is known as gelatinisation.
Flavor Concentration: Heating sauces causes water to evaporate, concentrating the flavors and thickening the sauce. This is why simmering a sauce can make it taste richer and more intense.
Baking Bread: When baking bread, heat causes the dough to rise as yeast ferments sugars, producing carbon dioxide. Meanwhile, the Maillard reaction and caramelization give the crust its golden-brown color and complex flavours.

Protein Transformation and Chemical Reactions
Cooking is more than just following recipes. It’s a dance between food and chemistry. Protein transformation is a key part of this dance. When heat is applied, proteins change their structure, affecting their properties.
Take eggs as an example. When you cook an egg, the heat causes the proteins to unfold and the egg white solidify. This changes the egg from clear and runny to white and firm. This change is due to protein denaturation, happening in many cooking methods.

Knowing about protein transformation helps chefs and home cooks. It lets them control the texture and flavour of their dishes. Whether making a soufflé or searing a steak, understanding protein chemistry can improve your cooking.
So, when you cook, think of yourself as a chemist. Explore the world of protein transformation. Cooking is not just about recipes. It’s about unlocking the potential of ingredients through chemistry.
Emulsification: Blending Oil and Water
Emulsification is the process of mixing two liquids that usually don’t combine, like oil and water. This is essential for making dressings, sauces, and mayonnaise.

An emulsifier, like the lecithin in egg yolks, helps stabilize the mixture by surrounding oil droplets and keeping them suspended in water.

When making vinaigrette, slowly whisking in the oil while stirring helps create a stable emulsion, resulting in a smooth and creamy dressing.
pH in Cooking
The acidity or alkalinity of ingredients can significantly impact the taste and texture of your dishes
Acidic Ingredients: Lemon juice (citric acid) or vinegar (acetic acid) can tenderize meat and add brightness to flavors.
They’re also used to prevent browning in fruits like apples and avocados.

Alkaline Ingredients: Baking soda (Sodium hydrogen carbonate) is an alkaline ingredient that helps dough rise by reacting with acidic components, releasing carbon dioxide.

How Chemical Reactions Create Texture and Flavour in Baking
Baking and creating pastries are really fascinating areas of food science. They reveal a world of chemical changes that create the textures, flavours, and structures we love in baked goods.
For example, when you mix the ingredients for a cake, the chemical reactions that occur as it bakes are what make the cake rise and develop its soft, fluffy texture. Or think about how a perfectly flaky pie crust is the result of specific chemical processes taking place in the oven.
Baking and pastry-making are like mini science experiments – you get to see the chemistry of cooking unfold right before your eyes!
Leavening Agents and Their Chemical Effects
Leavening agents like yeast and baking powder (sodium hydrogen carbonate) are key in baking chemistry. They cause dough or batter to rise, making baked products light and airy. Knowing how these agents work helps bakers control the final texture and rise.
Sugar’s Role in Texture and Structure
Sugar does more than add sweetness; it affects the pastry science of baking. It changes how proteins and starches work, influencing the texture and structure of baked goods.

By choosing the right sugar, bakers can control the chewiness, crispness, and colour of their creations.
Protein Networks in Dough Development
The formation of gluten is vital in dough development. Gluten, a protein network, traps gas from leavening agents. This baking chemistry helps dough rise and keep its shape, creating the right texture and crumb in breads and other baked goods.
Grasping these basic molecular processes lets bakers control their ingredients and techniques. This way, they can make consistently great pastry science creations. By using chemistry, bakers can explore new flavours, textures, and possibilities in baking and pastry making.

The Effects of Salt and Resting Meat
Salt (sodium chloride) is a crucial ingredient in cooking, enhancing flavour and affecting the texture of food. It helps draw out moisture and can improve the tenderness of meat by breaking down muscle fibres.
Resting meat after cooking allows the juices to redistribute, resulting in a juicier and more flavourful dish. This resting period ensures that when you cut into the meat, the juices remain inside rather than spilling out onto your plate.
Flavour Chemistry: Esters, Aldehydes, and Ketones
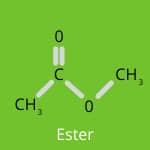
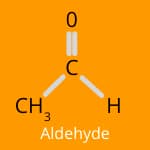
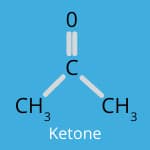
When we cook, the magical transformation of ingredients through heat and reactions like the Maillard reaction and caramelisation doesn’t just change the appearance of food—it also creates a beautiful blend of flavours.
This is largely due to the formation of various chemical compounds, including esters, aldehydes, and ketones, each contributing unique characteristics to the taste profile of our dishes.

Esters are often associated with fruity and floral aromas. They are responsible for the sweet, pleasant scents found in fruits like apples and strawberries. In cooking, esters can enhance the overall aroma of baked goods and desserts, adding subtle layers of sweetness and complexity.
Aldehydes bring a range of flavors, from sweet and nutty to green and grassy. They are prominent in the aroma of freshly cut grass and are also found in cinnamon and vanilla. In cooked foods, aldehydes can add depth to the flavour profile, making dishes more delicious and inviting.
Ketones are known for their creamy and buttery characteristics.
They are often found in dairy products and contribute to the rich flavours in cheese and butter. When foods are cooked, ketones can create a smooth, velvety taste that enhances the mouthfeel and richness of a dish.
Together, these compounds interact to create the complex and layered flavours we experience when eating cooked foods.

Understanding the role of these chemical groups can help aspiring chefs and home cooks appreciate the art and science of flavor creation, allowing them to experiment and enhance their culinary creations.
Conclusion
The world of food science is full of amazing changes in your kitchen. The Maillard reaction gives roasted meats their delicious flavours. It’s all about the chemistry of cooking.
By learning about food chemistry, you can improve your cooking. You’ll solve problems and make new dishes that excite your taste buds.
Whether you love cooking at home or do it professionally, exploring culinary chemistry is key. It opens up new ways to be creative and master your craft. As you learn more, you’ll see how chemistry makes food amazing.
Exploring the chemistry of cooking is endlessly fascinating. It lets you understand the science behind your favourite dishes. This knowledge opens up new possibilities in your kitchen.
So, dive into the world of food chemistry. It’s a journey of discovery and flavour. You’ll create meals that are not just delicious but also nourishing.
FAQ:
What is the relationship between chemistry and cooking?
Cooking is all about science, especially chemistry. It mixes science with creativity to make tasty meals. Knowing cooking chemistry helps chefs improve, from cooking scallops to making fermented foods.
What are the essential chemical components in cooking?
Key chemicals in cooking are proteins, carbohydrates, and fats. Salt, also boosts flavours.
How do chemical reactions transform raw ingredients into edible dishes?
Cooking chemistry changes raw ingredients into food we can eat. This includes protein changes, Maillard reactions, and caramelisation. Heat, acids, and enzymes are key in these changes.
What are the main methods of heat transfer in cooking?
There are three main ways heat transfers in cooking. Conduction is direct heat, Convection uses air or water, and Radiation uses waves. Each method affects how food cooks.
How does protein transformation affect the texture and flavour of cooked food?
Protein changes are vital in cooking. Heat makes proteins change, affecting texture and taste. The Maillard reaction, for example, browns and flavours food.
What is the Maillard reaction, and how does it affect food?
The Maillard reaction happens when proteins and sugars heat up. It browns and flavours food. Unlike caramelisation, it doesn’t just break down sugars. It’s all about the right heat for the perfect colour.
How does the chemistry of baking transform raw ingredients into pastries and breads?
Baking changes ingredients into tasty treats. Yeast and baking powder create air pockets. Sugar adds sweetness and changes texture. Gluten in dough gives bread its texture.



0 Comments